All categories

First Aid Kit
-
Humanized Service
-
Excellent post-purchase
-
Quality products
First Aid Kit
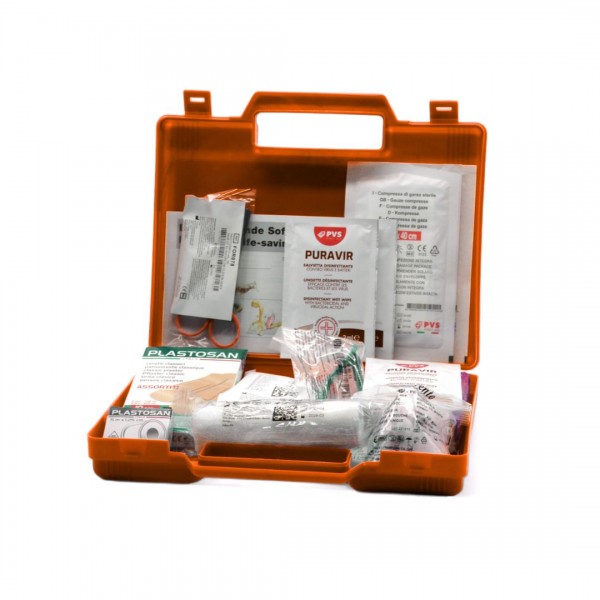
This first aid kit is ideal for treating minor injuries. Contains a polypropylene box, a handle for easy transport and 2 snap closures. It is a good option to carry with you and have in professional or personal environments.
What's in this first aid box?
2 sterile gauze bags 18×40 cm;
1 pair of scissors;
1 dressing tweezers;
1 sterile sheet 60×40 cm;
1 VENTO mouth to mouth resuscitator
1 pack from 10 plasters;
1 packet of paper handkerchiefs ;
3 disinfectant wipes;
1 elasticated bandage 4×10 cm;
1 adhesive bandage 10×6 cm;
1 roll of plaster m 5×1.25 cm;
1 pair of sterile protective gloves.
Dimensions
235x195x45 mm
Weight: 0.3kg
Empty box weight: 0.15 kg
Note: it is important to carry out a periodic review of the utensils and disinfectants to ensure that they are in perfect condition for use and within the warranty. Replace used materials as soon as possible.




 0/5
0/5

 Reviews
Reviews